Adlai Nortye Reports Full Year 2024 Financial Results and Business Updates
SINGAPORE and NORTH BRUNSWICK, N.J. and HANGZHOU, China, April. 30, 2025 -- Adlai Nortye Ltd. (NASDAQ: ANL) (the "Company" or "Adlai Nortye"), a global clinical-stage biotechnology company focused on the development of innovative targeted and immune-modulating cancer therapies, today announced its financial results for the full year ended December 31, 2024, along with a corporate update on key pipeline developments.
"We are proud of the meaningful progress across our pipeline of targeted and immune-modulating therapies," said Carsten Lu, CEO and Chairman of Adlai Nortye. "We are also highly encouraged by the progress of AN8025 and AN9025, which were highlighted at the 2025 AACR Annual Meeting. In addition, our lead candidate, AN2025, will remain on track to report overall survival data of its Phase 3 study in the first half of 2025."
Pipeline Updates
AN2025: pan-PI3K inhibitor
- The Company is conducting BURAN (NCT04338399), a randomized, open-label phase 3 trial to assess the treatment effect of once-daily buparlisib in combination with weekly paclitaxel compared to weekly paclitaxel alone in patients with recurrent or metastatic HNSCC that has progressed after prior anti-PD(L)1 treatment.
- Overall survival (OS) data to be presented in the first half of 2025.
AN4005: oral small-molecule PD-L1 inhibitor
- The Phase 1 trial of AN4005 (NCT04999384) is conducting dose expansion to evaluate two doses of AN4005 in checkpoint inhibitor-naïve patients.
- Preliminary results from the dose escalation were presented at SITC 2024, where AN4005 exhibits favorable safety profile. Preliminary efficacy was observed in a tumor type known to respond to anti-PD-(L)1 therapy, supporting further development.
AN8025: tri-specific T cell and APC modulator (αPD-L1/Lag3v/CD86v)
- Preclinical results of AN8025 was presented at the 2025 annual meeting of the American Associated for Cancer Research ("AACR"), with the poster title as "Coordinated stimulation of APC and T cell functions by first-in-class, tri-specific AN8025 for next-generation cancer immunotherapy".
- IND submission of AN8025 is anticipated by mid-2025
AN9025: oral small molecule pan-RAS(ON) inhibitor
- Preclinical results of AN9025 was presented at the 2025 AACR annual meeting, with the poster title as "AN9025, an orally bioavailable pan-RAS(ON) inhibitor with potent, broad-spectrum anti-tumor activity"
- IND submission of AN9025 is anticipated in the second half of 2025.
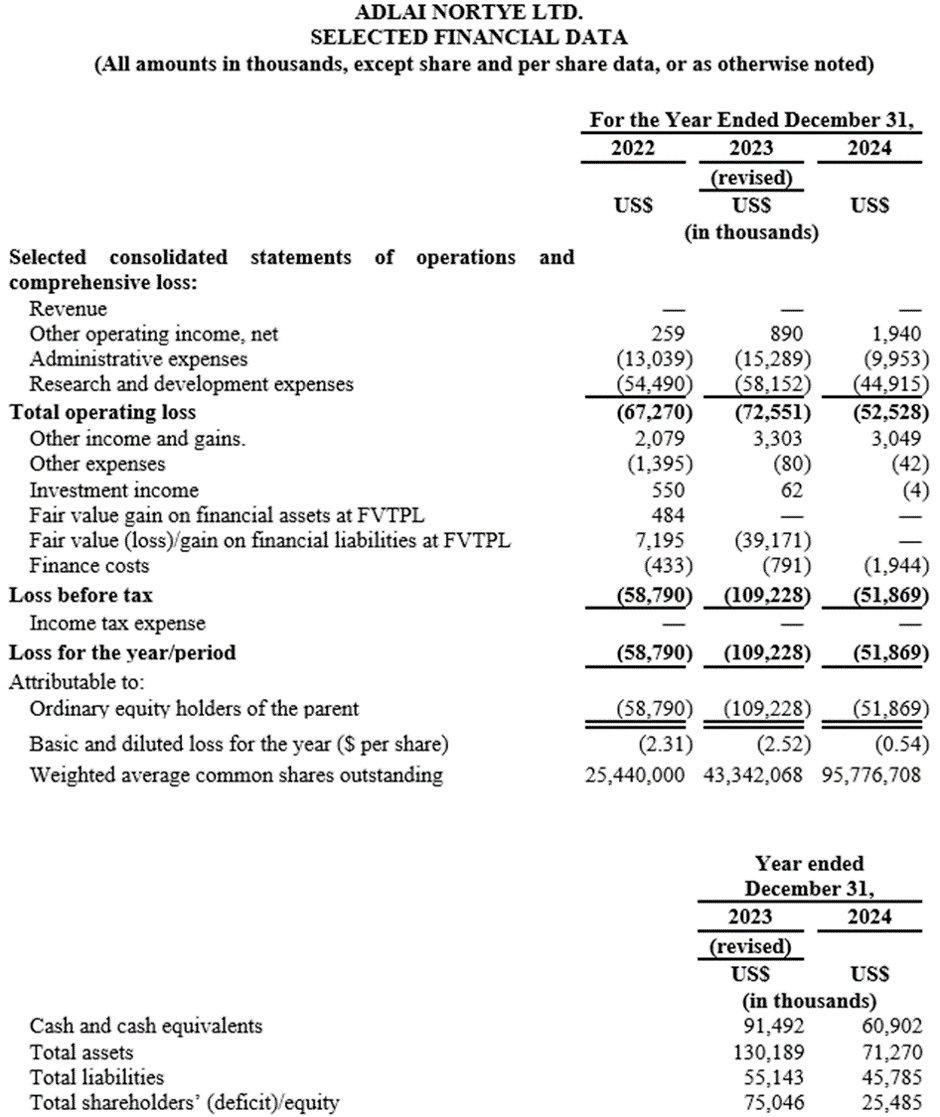
Full Year 2024 Financial Highlights
Research and development expenses decreased by 22.8%, from US$58.2 million in 2023 to US$44.9 million in 2024, primarily attributable to (i) a decrease of US$10.4 million in the CRO service fees as the completion of patient enrollment for the Phase III clinical trial of project AN2025 in 2023, only relevant clinical operational expenses were incurred in 2024, with no material other CRO service fees required thereafter and (ii) a decrease of US$2.8 million in the payroll expenses as the downsizing of U.S based R&D personnel.
Administrative expenses decreased by 34.9%, from US$15.3 million in 2023 to US$10.0 million in 2024, primarily attributable to (i) a decrease in IPO related professional service expenses in 2024. (ii) a decrease in share-based compensation expenses resulting from the vesting schedule of certain stock options in 2024.
Other income and gains decreased by 7.7% from US$3.3 million in 2023 to US$3.0 million in 2024, primarily attributable to a moderate reduction in government grants received during the year 2024.
Fair value loss on financial liabilities at FVTPL of US$39.2 million in 2023 and fair value loss on financial liabilities at FVTPL of US$0 in 2024. This change was primarily due to all of the financial liabilities at FVTPL converted into ordinary shares after IPO with no fair value loss on financial liabilities at FVTPL incurred thereafter.
Loss for the year decreased from US$109.2 (revised) million in 2023 to US$51.9 million in 2024.
As of December 31, 2024, the Company had US$60.9 million in cash and cash equivalents, compared to US$91.5 million for the year ended December 31, 2023.
About Adlai Nortye
Adlai Nortye (NASDAQ: ANL) is a global clinical-stage company focused on the development of innovative targeted and immune-modulating cancer therapies, with global R&D centers in the U.S. and China. The Company is advancing a robust oncology pipeline, with our lead candidate Buparlisib (AN2025), a pan-PI3K inhibitor, currently being evaluated in a registrational Phase 3 trial (NCT04338399) in patients with recurrent or metastatic head and neck squamous cell cancer (HNSCC) that has progressed after prior anti-PD(L)1 treatment. Additionally, we are advancing multiple drug candidates including AN4005, an oral small molecule PD-L1 inhibitor, AN8025, a multifunctional fusion protein acting as a T cell and antigen-presenting cell modulator (αPD-L1/Lag3v/CD86v), and AN9025, an oral small molecule pan-RAS(ON) inhibitor.
Forward-Looking Statement
This announcement contains forward-looking statements. These statements are made under the “safe harbor” provisions of the U.S. Private Securities Litigation Reform Act of 1995. These forward-looking statements can be identified by terminology such as “will,” “expects,” “anticipates,” “future,” “intends,” “plans,” “believes,” “estimates,” “potential,” “continue,” “ongoing,” “targets” and similar statements. Among other things, statements that are not historical facts, including statements about the Company’s beliefs and expectations, the business outlook and quotations from management in this announcement, as well as the Company’s strategic and operational plans, are or contain forward-looking statements.
The Company may also make written or oral forward-looking statements in its periodic reports to the U.S. Securities and Exchange Commission (the “SEC”), in press releases and other written materials and in oral statements made by its officers, directors or employees to third parties. Forward-looking statements involve inherent risks and uncertainties. Factors that could cause the Company’s actual results to differ materially from those expressed or implied in such forward-looking statements include, but are not limited to: the initiation, timing, progress and results of the Company’s preclinical studies, clinical trials and other therapeutic candidate development efforts; the Company’s ability to advance its therapeutic candidates into clinical trials or to successfully complete its preclinical studies or clinical trials; whether the clinical trial results will be predictive of real-world results; the Company’s receipt of regulatory approvals for its therapeutic candidates, and the timing of other regulatory filings and approvals; the clinical development, commercialization and market acceptance of the Company’s therapeutic candidates; the Company’s ability to establish, manage, and maintain corporate collaborations, as well as the ability of its collaborators to execute on their development and commercialization plans; the implementation of the Company’s business model and strategic plans for its business and therapeutic candidates; the scope of protection the Company is able to establish and maintain for intellectual property rights covering its therapeutic candidates and its ability to operate its business without infringing the intellectual property rights of others; estimates of the Company’s expenses, future revenues, capital requirements and its needs for and ability to access sufficient additional financing; risks related to changes in healthcare laws, rules and regulations in the PRC and United States or elsewhere. Further information regarding these and other risks is included in the Company’s filings with the SEC. All information provided in this announcement and in the attachments is as of the date of this announcement, and the Company does not undertake any obligation to update any forward-looking statement, except as required under applicable law.